Cells move by controlling the stiffness of their neighbors
New insight on how cells migrate through the body could have implications for developing new ways to slow or prevent the spread of cancer.
An unexpected finding shows that cells moving through tissue can facilitate their own migration by altering the stiffness of the surrounding cells. The findings from Tina Tootle’s lab at the University of Iowa Carver College of Medicine have implications for understanding problems caused by abnormal cell migration, like birth defects and cancer metastasis.
Previous laboratory studies of cell migration have shown that cells move more quickly and easily across stiffer surfaces, but much of this research has been done using artificial surfaces. In the new study, published in the journal eLife, Tootle’s team, including lead author Maureen Lamb, investigated the process using a natural tissue environment.
The team examined the movement of cells in the ovaries of fruit flies. They looked specifically at what happened in cells that were genetically engineered to lack a protein called fascin, which regulates a cell-stiffening protein called myosin.
“What we found was not what we expected,” says Tootle, PhD, an associate professor of anatomy and cell biology in the UI Carver College of Medicine and senior study author. “We expected that the stiffness of the surrounding tissue would control migratory cell movement. What we found is the migratory cells are driving changes in their surrounding tissue to promote their own migration.”
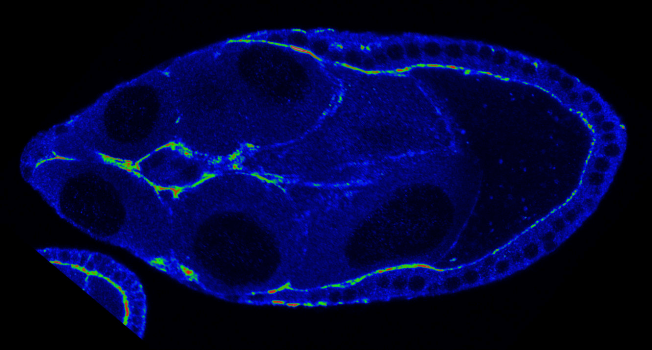
In collaboration with colleagues in the UI Department of Chemistry, Tootle’s team used a technique called atomic force microscopy to measure the stiffness of the cells. They found that in the cells lacking fascin, the activity of myosin increased, and both the migrating cells and their neighboring cells stiffened, hindering their movement. When the team restored fascin to the migrating cells, this led to normal myosin activity and stiffness of the neighboring cells, which promoted cell movement. They also discovered a mechanism that cells can use to turn fascin on and off.
“We’ve shown that migrating cells use fascin to control the stiffness of surrounding cells, making it easier for them to move through the body,” say Lamb, who was a UI graduate student in Tootle’s lab and is now at the Stowers Institute for Medical Research in Kansas City.
The surprising discovery that cells can control their ability to move by regulating the stiffness of surrounding cells may have important implications for understanding how cancer cells spread. Cancer that has metastasized and spread beyond the original tumor is often more difficult to treat and reduces the patient’s chances of survival.
“If cancer cells use the same mechanisms identified in our study, then it may be possible to reduce their spread by altering the tissues that they prefer to move across, so that the tissues don’t stiffen in response to the cells,” say Tootle, who also is a member of Holden Comprehensive Cancer Center at the UI. “The next step is to test this idea in larger organisms to see if it could be a potential strategy to slow or prevent the spread of cancer.”
In addition to Tootle and Lamb, the UI research team included Chathuri Kaluarachchi, Thiranjeewa Lansakara, Samuel Mellentine, Yiling Lan, and Alexei Tivanski. The study was funded in part by a grant from the National Institute of General Medical Sciences.