A new study identifies a single molecule as a key entry point used by two types of dangerous bacteria to break through cellular barriers and cause disease. The findings, published March 19 in the journal mBio, suggest that blocking the interaction between the molecule, known as CD40, and bacteria may represent a universal strategy for preventing life-threatening illnesses, including toxic shock syndrome.
Different bacteria use same cell surface molecule to invade tissue
The two bacteria, Staphylococcus aureus (staph) and Streptococcus pyogenes, cause many serious illnesses. According to the Centers for Disease Control and Prevention, each year staph causes 70,000 cases of highly fatal pneumonia, 40,000 cases of severe heart infections, and more than 500,000 post-surgical infections. Streptococcus pyogenes annually causes 10 million cases of sore throat and 30,000 cases of severe invasive diseases.

Previous work by Schlievert showed that superantigens cause the epithelial cells of the mucosal barrier to make signaling molecules called chemokines. Those chemokines attract immune cells that disrupt the integrity of the mucosal barrier, allowing the superantigens and frequently the bacteria themselves to penetrate and cause serious and often fatal diseases, including toxic shock syndrome.
In the new study, Schlievert and his colleagues at the UI and National Jewish Health in Denver used human vaginal epithelial cells as a model of a mucosal barrier. They showed that the interaction between CD40 and superantigens triggers the production of chemokines in these cells. Removing CD40 from the cells using CRISPR gene editing prevented bacterial superantigens from triggering the production of the chemokines. In contrast, restoring CD40 to those cells restored the ability of superantigens to trigger chemokine production.
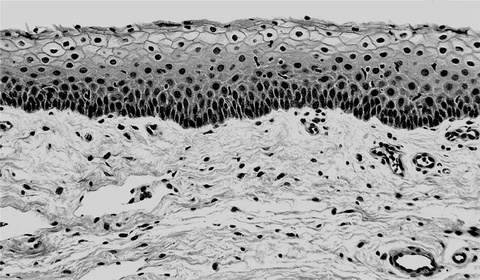
The human vaginal mucosa (thick, dark layer of cells at the top of the picture) is a cellular barrier that physically prevents pathogens and dirt from entering the body. Bacterial toxins called superantigens interact with CD40 molecules on the mucosa surface and trigger chemokine production. This leads to disruption of the barrier and allows the superantigens and in many cases the bacteria themselves to penetrate and causes serious and often fatal diseases, including toxic shock syndrome.
Blocking bacteria-molecule interaction might prevent many deadly illnesses
The team tested three superantigens: toxic shock syndrome toxin 1 (TSST-1), and staphylococcal enterotoxin B and C. Of the three, TSST-1 produced the strongest effect, which likely explains why this superantigen is responsible for all cases of menstrual toxic shock syndrome.
“CD40 is critical; you get no response (to the superantigens) without it,” Schlievert says. “Since we now know that these two large families of bacteria cause disease through the same human tissue receptor, we are looking for ways to block the interaction between CD40 and superantigens to prevent immune activation. This work also raises the possibility that many other pathogens may cause diseases through the same receptor interaction. Those studies are underway.
“If there is a central pathway that many pathogens use to disrupt mucosal barriers, that would mean we have a single target to block to prevent barrier disruption by pathogens. That could be huge,” he adds.
Members of the research team
In addition to Schlievert, the research team included Michael Cahill, Bruce Hostager, Aloysius Klingelhutz, Francoise Gourronc, and Gail Bishop, all from the University of Iowa; Amanda Brosnahan from the University of Minnesota; and Donald Y. M. Leung from National Jewish Health in Denver.
The study was funded in part by grants from the National Institutes of Health.